When nothing else works, desperate clinicians are resorting to progressively more-tenuous and unpredictable treatments, trying to improve the lives of patients with refractory schizophrenia. High-dose antipsychotics is a common strategy.
Does boosting antipsychotic doses beyond the recommended range—but short of the neuroleptic threshold—enhance efficacy? This article attempts to answer that question by presenting the evidence on higher-than-recommended doses of atypical antipsychotics.
Lessons from neuroleptics
Up to 30% of patients with schizophrenia do not respond to antipsychotics and are considered “treatment refractory.”1 Even among those who do respond, improving symptoms by 20%—as research defines “treatment response”—does not necessarily yield clinical or functional improvement. Clozapine is the only atypical antipsychotic with well-established efficacy in these chronically ill patients,2 but its daunting side effects greatly curtail its use.
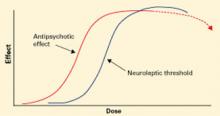
Before atypical antipsychotics, patients who did not respond to usual dosages of the typical neuroleptics were treated with higher dosages or switched to another drug class. Although many clinicians embraced high-dose neuroleptics, subsequent research discredited “rapid neuroleptization” in any clinical circumstance and showed that exceeding an antipsychotic’s neuroleptic threshold—the dose at which extrapyramidal side effects (EPS) occur—reduces its efficacy (Figure 1).3-5 In some instances, reducing neuroleptic dosages improves treatment-resistant patients’ symptoms and reduces druginduced side effects.6
Figure 1 Typical antipsychotics’ dose-response curve
Narrow therapeutic window between antipsychotic effect and neuroleptic threshold. Dotted line indicates declining efficacy.
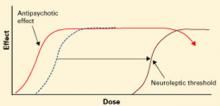
Figure 2 Atypical antipsychotics’ dose-response curve
Wider therapeutic window with atypicals, compared with typical antipsychotics, as neuroleptic threshold (dotted line) moves right.Atypical antipsychotics are defined by their relative lack of EPS at recommended dosages (Figure 2). Because these agents can cause EPS if dosed too high, however, our historical habit of testing this dose limit risks losing “atypicality” and encountering other untoward events (Figure 3).
What is the safest, most effective dosage? Consider the evidence for each atypical antipsychotic.
Risperidone
Recommended dosage too high? When using atypicals at recommended doses, you are most likely to encounter the neuroleptic threshold with risperidone, with EPS risk increasing substantially at >6 mg/d.7 Post-approval studies set the most effective and safest dosage at approximately 4 mg/d, though this dosage was not studied in North American pre-approval trials. Dosages of 2 to 4 mg/d have been associated with more-favorable outcomes, suggesting that the initial recommendation to titrate to 6 mg/d within the first 3 days was ill-advised.8
In our study of patients with treatment-refractory schizophrenia,9 those treated with risperidone, 6 mg/d, improved significantly more after 4 weeks than did those receiving haloperidol, 15 mg/d, based on Brief Psychiatric Rating Scale (BPRS) scores. No additional benefit was seen after risperidone was increased to >6 mg/d at 8 weeks. Akathisia and tardive dyskinesia occurred significantly more often in the haloperidol group.
Conclusion. Some patients respond to higher-dose risperidone, but emerging EPS suggest the need to reduce the dosage rather than add an antiparkinsonian agent.
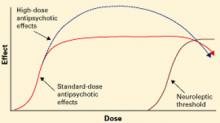
Figure 3 Unknown effects of high-dose atypical antipsychotic therapy
Dotted line indicates potential for greater antipsychotic effect with increasing dose.
Olanzapine
Mixed results. Case reports suggest that some patients who did not respond to previous antipsychotic trials or olanzapine, 20 mg/d, improved sig-nificantly—without substanial side effects—when olanzapine was increased up to 60 mg/d.10-14 Other case studies, however, report EPS, increased heart rate, increased transaminases, hyperprolactinemia, and prolonged QTc interval with high-dose olanzapine.14-16
In an open-label trial,17 43 patients with schizophrenia received olanzapine, up to 40 mg/d, after inadequate response to neuroleptics and risperidone or clozapine. Olanzapine was titrated to 20 mg/d by week 4 and increased 5 mg every 2 weeks if symptoms did not improve. After 14 weeks, improvement was modest and only 17% of patients met response criteria. However, >20 mg/d reduced symptoms more than did <20 mg/d, suggesting that high-dose olanzapine was more effective.
In a randomized trial,18 patients who did not respond to at least one atypical antipsychotic then received 8 weeks of fixed, standard-dose treatment with (mean dosages):
- haloperidol, 18.9 mg/d
- risperidone, 7.9 mg/d
- olanzapine, 19.6 mg/d
- clozapine, 401.6 mg/d.
Flexible dosing was then allowed for 6 weeks, and mean dosages were:
- haloperidol, 25.7 mg/d
- risperidone, 11.6 mg/d
- olanzapine, 30.4 mg/d
- clozapine, 526.6 mg/d.
Symptoms improved modestly at best for all medications, although patients taking olanzapine or clozapine improved significantly more than those treated with haloperidol as shown by mean changes in total Positive and Negative Syndrome Scale (PANSS) scores.
PANSS scores for olanzapine-treated patients showed additional improvement at week 14—when higher dosages were used—compared with week 8. This was not the case for the other medications, for which response plateaued. These findings suggest that high-dose risperidone and haloperidol are incrementally ineffective, but high-dose olanzapine could help some patients with refractory symptoms.